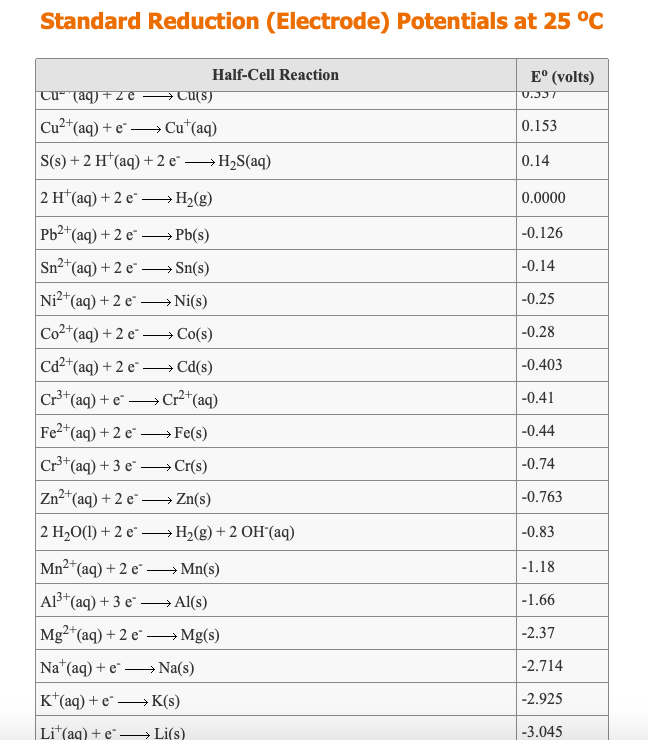
Using the standard electrode potentials given in the Table, predict the reaction between the following is feasible.Ag(s) and Fe^{3+}(aq)

Table 4 from Estimation of standard reduction potentials of halogen atoms and alkyl halides. | Semantic Scholar

electrochemistry - How to calculate whether a redox reaction is spontanuous? - Chemistry Stack Exchange

Table 1 from Absolute standard hydrogen electrode potential measured by reduction of aqueous nanodrops in the gas phase. | Semantic Scholar

SOLVED: Table A5.5 Standard Reduction Potentials at 25°C (298 K) for Many Common Half-Reactions Half-Reaction Half-Reaction 4OH- + F2 â†' 2F- + 2H2O 2.87 2H2O + 4e- â†' Cu2+ + 2OH- 0.01



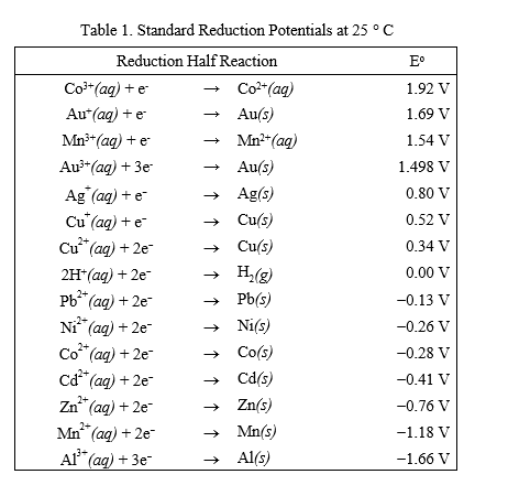



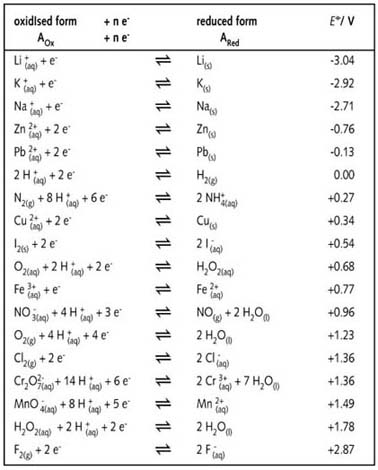

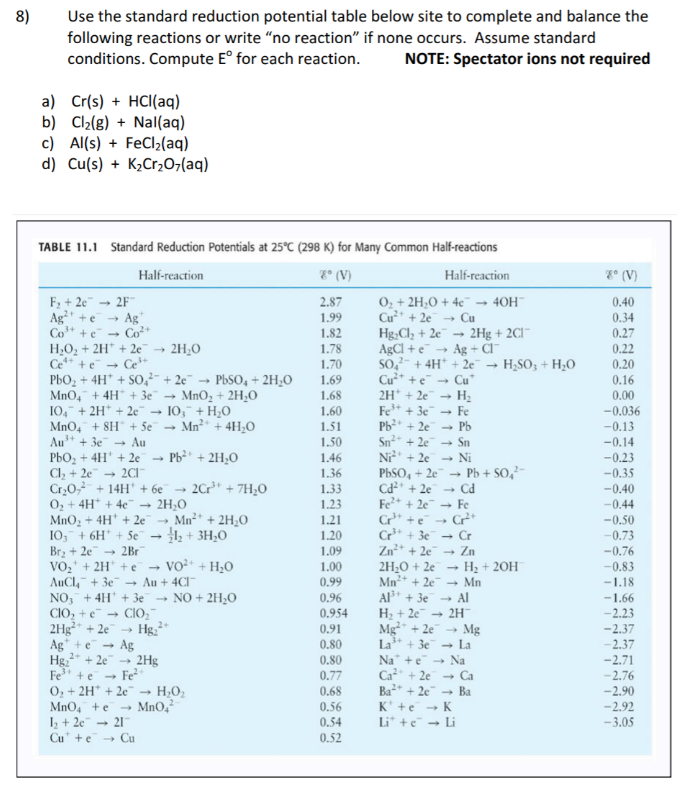








![Standard reduction potentials at 298°K. [24] | Download Table Standard reduction potentials at 298°K. [24] | Download Table](https://www.researchgate.net/publication/316026333/figure/tbl2/AS:650784626708491@1532170554986/Standard-reduction-potentials-at-298K-24.png)